For decades, the sense of smell has been the outlier in neuroscience. While we have detailed maps explaining how our eyes process light, our ears interpret sound, and our skin registers touch, olfaction remained a scientific black box. Researchers long assumed that smell-sensing neurons were scattered randomly throughout the nose, a chaotic arrangement that made it difficult to understand how the brain organizes scent information.
That assumption has now been overturned.
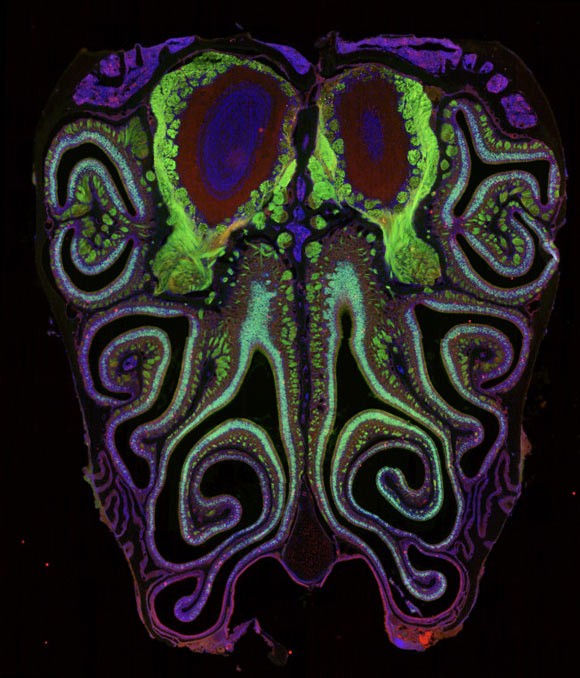
In a groundbreaking study published in Cell, researchers from Harvard Medical School and colleagues have created the first high-resolution map of smell receptors in the mouse nose. Their findings reveal that olfactory neurons are not randomly distributed but are organized into precise, horizontal stripes. This discovery not only solves a long-standing mystery but also provides a critical roadmap for developing treatments for smell loss, a condition that currently lacks effective therapies.
From Chaos to Order
Led by Professor Sandeep (Robert) Datta, the research team challenged the prevailing view that olfactory receptor neurons were disorganized. To prove this, they needed to look at the system at an unprecedented scale.
The team analyzed approximately 5.5 million neurons across more than 300 mice. This massive dataset represents arguably the most sequenced neural tissue ever examined. By combining two advanced techniques—single-cell sequencing and spatial transcriptomics —the scientists achieved a dual insight:
1. They identified exactly which smell receptors each neuron expressed.
2. They pinpointed the exact physical location of those neurons within the nose.
The result was striking. Rather than a random scatter, the neurons formed tight, overlapping horizontal stripes running from the top to the bottom of the nasal cavity. Crucially, these stripes were organized by receptor type. Neurons expressing the same type of smell receptor clustered together in specific bands.
A Bridge to the Brain
The true significance of this nasal map lies in its connection to the brain. In other senses, such as vision and hearing, there is a clear spatial correspondence between the sensory organ and the brain’s processing centers. For example, specific parts of the retina map directly to specific parts of the visual cortex.
The new study shows that olfaction follows the same rule. The striped map in the nose mirrors the organization of smell maps in the olfactory bulb of the brain. This alignment suggests a highly efficient, logical pathway for how scent information travels from the nose to the brain, overturning the idea that smell processing is inherently disordered.
“Maps have long existed that describe how receptors in the eye, ear, and skin are organized… However, olfaction has been the one exception; it’s the sense that has been missing a map for the longest time.”
— Professor Sandeep Datta
How the Map Forms: The Role of Retinoic Acid
Understanding what the map looks like is only half the story. The researchers also investigated how this complex organization develops during embryonic growth. How does the nose know to place specific receptors in specific stripes?
The answer lies in a molecule called retinoic acid, which helps control gene activity. The team discovered that retinoic acid forms a gradient within the developing nose—meaning its concentration varies gradually from one end to the other.
This chemical gradient acts as a spatial guide:
* Neurons sense their position based on the local concentration of retinoic acid.
* This signal instructs each neuron which specific smell receptor to express.
* The result is a self-organizing system that consistently produces the same striped pattern across different animals.
To prove this mechanism, the researchers manipulated the levels of retinoic acid. Adding or removing the molecule caused the receptor map to shift up or down, confirming that this chemical gradient is the key driver of olfactory organization.
Why This Matters
This discovery is more than just a biological curiosity; it has profound implications for human health. Loss of smell (anosmia) affects millions of people and can be an early indicator of neurological disorders like Parkinson’s and Alzheimer’s disease. Yet, effective treatments remain elusive because the underlying wiring of the sense of smell was poorly understood.
“We cannot fix smell without understanding how it works on a basic level,” Professor Datta noted. By revealing the structural logic of olfaction, this study provides the foundational knowledge necessary to develop targeted therapies. It transforms smell from a mysterious, uncharted sense into a mapped system that can be studied, understood, and potentially repaired.
Conclusion
The mapping of smell receptors in the mouse nose resolves a decades-old puzzle in neuroscience, revealing that olfaction is as structured and logical as vision or hearing. By identifying the role of retinoic acid in creating this precise spatial code, scientists have not only explained how smell works but also opened new avenues for treating smell-related disorders.